| NCT01715584 | Hypertension | Phase 4 | Recruiting | December 31, 2019 | Canada, Ontario
... more >>
London Health Sciences Centre - Victoria Campus
Recruiting
London, Ontario, Canada, N6A 5W9
Contact: Craig J Railton, MD, PhD 519 685 8500 ext 58525 Craig.Railton@lhsc.on.ca
Principal Investigator: Craig J Railton, MD, PhD
Sub-Investigator: Jonathan Fairbairn, BSc
Sub-Investigator: George Nicoloau, MD
Sub-Investigator: Robert Gros, PhD
Sub-Investigator: Jason Franklin, MD
Sub-Investigator: John Yoo, MD
Sub-Investigator: Kevin Fung, MD
Sub-Investigator: Anthony Nichols, MD
Sub-Investigator: Danielle McNeil, MD Collapse << |
| NCT02235909 | Hypertension | Phase 3 | Recruiting | August 2020 | - |
| NCT00362115 | Hypertension | Phase 2 | Completed | - | - |
| NCT03006796 | - | - | Recruiting | June 2019 | Russian Federation
... more >>
City Hospital №5
Recruiting
Moscow, Russian Federation, 107015
Contact: Elena Y Shupenina, PhD +74992684459 Collapse << |
| NCT00759551 | Hypertension | Phase 2 | Completed | - | - |
| NCT00362115 | - | - | Completed | - | - |
| NCT00762736 | Diabetes Mellitus | Phase 2 | Completed | - | - |
| NCT00376181 | Type 2 Diabetes | Phase 3 | Terminated(Combination formula... more >>tion concerns) Collapse << | - | - |
| NCT00696436 | Hypertension | Phase 3 | Completed | - | - |
| NCT00847626 | - | - | Completed | - | - |
| NCT00847626 | Hypertension | Phase 3 | Completed | - | - |
| NCT01289132 | Essential Hypertension | Phase 2 | Completed | - | - |
| NCT02100319 | - | - | Completed | - | Japan
... more >>
Osaka, Japan
Tokyo, Japan Collapse << |
| NCT00696241 | Hypertension | Phase 3 | Completed | - | - |
| NCT02348658 | Hypertension | Phase 1 | Completed | - | Japan
... more >>
Fukuoka-shi, Fukuoka, Japan Collapse << |
| NCT02348658 | - | - | Completed | - | - |
| NCT00696436 | - | - | Completed | - | - |
| NCT02401464 | - | - | Completed | - | - |
| NCT02401464 | Japanese Healthy Adult Males | Phase 1 | Completed | - | Japan
... more >>
Kagoshima, Japan Collapse << |
| NCT02277691 | Essential Hypertension | Phase 3 | Completed | - | Japan
... more >>
Nagoya-shi, Aichi, Japan
Chiba-shi, Chiba, Japan
Itojima-shi, Fukuoka, Japan
Kouriyama-shi, Fukushima, Japan
Sapporo-shi, Hokkaido, Japan
Amagasaki-shi, Hyougo, Japan
Tsukuba-shi, Ibaragi, Japan
Morioka-shi, Iwate, Japan
Sakaide-shi, Kagawa, Japan
Takamatsu-shi, Kagawa, Japan
Kawasaki-shi, Kanagawa, Japan
Kyoto-shi, Kyoto, Japan
Uji-shi, Kyoto, Japan
Sendai-shi, Miyagi, Japan
Hirakata-shi, Osaka, Japan
Osaka-shi, Osaka, Japan
Takatsuki-shi, Osaka, Japan
Saitama-shi, Saitama, Japan
Tokorozawa-shi, Saitama, Japan
Yaizu-shi, Shizuoka, Japan
Chiyoda-ku, Tokyo, Japan
Choufu-shi, Tokyo, Japan
Kodaira-shi, Tokyo, Japan
Koutou-ku, Tokyo, Japan
Setagaya-ku, Tokyo, Japan
Shinagawa-ku, Tokyo, Japan
Shinjuku-ku, Tokyo, Japan
Choufu-shi, Japan
Kawasaki-shi, Japan
Koutou-ku, Japan
Morioka-shi, Japan
Sakaide-shi, Japan
Setagaya-ku, Japan
Shinagawa-ku, Japan
Shinjuku-ku, Japan
Tsukuba-shi, Japan
Uji-shi, Japan
Yaizu-shi, Japan Collapse << |
| NCT00591266 | Hypertension | Phase 3 | Completed | - | - |
| NCT00696241 | - | - | Completed | - | - |
| NCT02277691 | - | - | Completed | - | - |
| NCT00591773 | Hypertension | Phase 3 | Completed | - | - |
| NCT00591266 | - | - | Completed | - | - |
| NCT00760214 | Hypertension | Phase 3 | Completed | - | - |
| NCT01124656 | Diabetes Mellitus, Type 2 | Phase 3 | Terminated(Formulation issues.... more >>) Collapse << | - | - |
| NCT00591773 | - | - | Completed | - | - |
| NCT00760214 | - | - | Completed | - | - |
| NCT01078376 | Hypertension | Phase 1 | Terminated(Business Decision (... more >>see below)) Collapse << | - | United States, Arkansas
... more >>
Little Rock, Arkansas, United States
United States, Georgia
Atlanta, Georgia, United States
United States, Kentucky
Louisville, Kentucky, United States
United States, Ohio
Toledo, Ohio, United States
United Kingdom
Birmingham, England, United Kingdom
Bristol, England, United Kingdom
London, England, United Kingdom
Manchester, England, United Kingdom Collapse << |
| NCT00696384 | Hypertension | Phase 3 | Completed | - | - |
| NCT01078376 | - | - | Terminated(Business Decision (... more >>see below)) Collapse << | - | - |
| NCT00696384 | - | - | Completed | - | - |
| NCT00846365 | - | - | Completed | - | - |
| NCT00591253 | Hypertension | Phase 3 | Completed | - | - |
| NCT00846365 | Essential Hypertension | Phase 3 | Completed | - | - |
| NCT01985152 | Health | Phase 1 | Unknown | - | China
... more >> TEDA International Cardiovascular Hospital
Recruiting
Tianjin, China
Contact: Jie Hou, Associate Chief Physician 86-022-65209939
Principal Investigator: Jie Hou, Associate Chief Physician Collapse << |
| NCT00591578 | Hypertension | Phase 3 | Completed | - | - |
| NCT00591578 | - | - | Completed | - | - |
| NCT02072330 | Grade I or II Essential Hypert... more >>ension Collapse << | Phase 2
Phase 3 | Completed | - | Japan
... more >>
Touon-shi, Ehime, Japan
Fukuoka-shi, Fukuoka, Japan
Hiroshima-shi, Hiroshima, Japan
Sapporo-shi, Hokkaido, Japan
Hanamaki-shi, Iwate, Japan
Morioka-shi, Iwate, Japan
Kumamoto-shi, Kumamoto, Japan
Kyoto-shi, Kyoto, Japan
Sendai-shi, Miyagi, Japan
Osaka-shi, Osaka, Japan
Suita-shi, Osaka, Japan
Shinjuku-ku, Tokyo, Japan
Toyama-shi, Toyama, Japan Collapse << |
| NCT00996281 | - | - | Completed | - | - |
| NCT01309828 | - | - | Completed | - | - |
| NCT00591253 | - | - | Completed | - | - |
| NCT00996281 | Essential Hypertension | Phase 3 | Completed | - | Austria
... more >>
Graz, Styria, Austria
Germany
Karlsruhe, Baden-Wurttemberg, Germany
Hannover, Lower Saxony, Germany
Kiel-Kronshagen, Schleswig-Holstein, Germany
Netherlands
Breda, North Brabant, Netherlands
Eindhoven, North Brabant, Netherlands
Amsterdam, North Holland, Netherlands
Velp, Rheden, Netherlands
Leiderdorp, South Holland, Netherlands
Zoetermeer, South Holland, Netherlands
Rotterdam, Zuid-Holland, Netherlands
Groningen, Netherlands
Poland
Bydgoszcz, Kuyavian-Pomeranian, Poland
Skierniewice, L0dz, Poland
Zgierz, L0dz, Poland
Gdansk, Pomeranian, Poland
Gdynia, Pomeranian, Poland
Sopot, Pomeranian, Poland
Mikolow, Silesian, Poland
United Kingdom
Avon, England, United Kingdom
Bolton, England, United Kingdom
Chorley, England, United Kingdom
Inverness, England, United Kingdom
Liverpool, England, United Kingdom
Surrey, England, United Kingdom
Warwickshire, England, United Kingdom Collapse << |
| NCT00695955 | Hypertension | Phase 3 | Completed | - | - |
| NCT01033071 | Essential Hypertension | Phase 3 | Completed | - | - |
| NCT01609959 | Hypertension | Phase 4 | Completed | - | Japan
... more >> First Department of Nara Medical University
Kashihara, Nara, Japan, 634-8522 Collapse << |
| NCT01033071 | - | - | Completed | - | - |
| NCT01309828 | Safety | Phase 3 | Completed | - | - |
| NCT00695955 | - | - | Completed | - | - |
| NCT02786082 | Hypertension | Phase 1 | Completed | - | - |
| NCT02079805 | Essential Hypertension Complic... more >>ated by Type 2 Diabetes Mellitus Collapse << | Phase 4 | Completed | - | Japan
... more >>
Kyoto-Shi, Kyoto, Japan Collapse << |
| NCT02079805 | - | - | Completed | - | - |
| NCT01496430 | Hypertension
... more >>Diabetes Collapse << | Phase 3 | Terminated(Business Decision; ... more >>No Safety Or Efficacy Concerns. (See below)) Collapse << | - | - |
| NCT02609490 | Hypertension | Phase 3 | Completed | - | China
... more >> Peking University First hospiatl
Beijing, China, 100000 Collapse << |
| NCT02407210 | Essential Hypertension | Phase 3 | Unknown | - | China
... more >> Beijing ANZHEN Hospital
Recruiting
Beijing, China, 100029
Contact: Changsheng Ma, Professor 86-010-64456078 chshma@vip.sina.com
Principal Investigator: Changsheng Ma, Professor Collapse << |
| NCT00818883 | Essential Hypertension | Phase 3 | Completed | - | - |
| NCT02181816 | - | - | Completed | - | Japan
... more >>
Osaka, Japan
Tokyo, Japan Collapse << |
| NCT01762501 | Hypertension | Not Applicable | Completed | - | - |
| NCT01762501 | - | - | Completed | - | - |
| NCT02541669 | Healthy Volunteer | Phase 1 | Completed | - | China, Beijing,P.R.
... more >>
Beijing, Beijing,P.R., China Collapse << |
| NCT00818883 | - | - | Completed | - | - |
| NCT03042299 | - | - | Completed | - | - |
| NCT01456169 | Essential Hypertension | Phase 3 | Completed | - | - |
| NCT02791438 | Pediatric Hypertension | Phase 3 | Active, not recruiting | June 11, 2019 | Japan
... more >>
Nagakute, Aichi, Japan
Nagoya, Aichi, Japan
Obu, Aichi, Japan
Sapporo, Hokkaido, Japan
Kobe, Hyogo, Japan
Kanazawa, Ishikawa, Japan
Yokohama, Kanagawa, Japan
Nankoku, Kochi, Japan
Sendai, Miyagi, Japan
Shimajiri-gun, Okinawa, Japan
Izumi, Osaka, Japan
Osaka Sayama, Osaka, Japan
Otsu, Shiga, Japan
Shimotsuke, Tochigi, Japan
Fuchu, Tokyo, Japan
Nerima-ku, Tokyo, Japan
Ota-ku, Tokyo, Japan
Setagaya-ku, Tokyo, Japan
Shinjuku-ku, Tokyo, Japan
Akita, Japan
Chiba, Japan
Fukuoka, Japan
Fukushima, Japan
Hiroshima, Japan
Niigata, Japan
Okayama, Japan
Osaka, Japan
Saitama, Japan
Shizuoka, Japan
Wakayama, Japan Collapse << |
| NCT01496430 | - | - | Terminated(Business Decision; ... more >>No Safety Or Efficacy Concerns. (See below)) Collapse << | - | - |
| NCT01456169 | - | - | Completed | - | - |
| NCT03434977 | Healthy Adult Male Participant... more >>s Collapse << | Phase 1 | Completed | - | Japan
... more >> Fukuoka Mirai Hospital
Fukuoka, Japan Collapse << |
| NCT02092025 | - | - | Completed | - | Japan
... more >>
Osaka, Japan
Tokyo, Japan Collapse << |
| NCT03042299 | Japanese Healthy Adult Male Pa... more >>rticipants Collapse << | Phase 1 | Completed | - | Japan
... more >> Nishi Kumamoto Hospital
Kumamoto, Japan Collapse << |
| NCT02480764 | Essential Hypertension | Phase 3 | Completed | - | - |
| NCT02203916 | - | - | Completed | - | - |
| NCT02400775 | Essential Hypertension With St... more >>able Angina and Dyslipidemia Collapse << | Phase 4 | Withdrawn | - | Japan
... more >>
Tokushima, Japan Collapse << |
| NCT03539627 | - | - | Not yet recruiting | June 2021 | - |
| NCT01774591 | Hypertension | Not Applicable | Completed | - | United States, Illinois
... more >>
University of Chicago
Chicago, Illinois, United States, 60637 Collapse << |
| NCT02203916 | Hypertension | Phase 3 | Completed | - | Korea, Republic of
... more >>
Chuncheon-Si, Gangwon-do, Korea, Republic of
Wonju-Si, Gangwon-do, Korea, Republic of
Anyang-si, Gyeonggi-do, Korea, Republic of
Goyang-si, Gyeonggi-do, Korea, Republic of
Seongnam-si, Gyeonggi-do, Korea, Republic of
Suwon-si, Gyeonggi-do, Korea, Republic of
Daegu, Gyeongsangbuk-do, Korea, Republic of
Yangsan-si, Gyeongsangnam-do, Korea, Republic of
Jeonju-si, Jeollabuk-do, Korea, Republic of
Gwangju, Jeollanam-do, Korea, Republic of
Busan, Korea, Republic of
Daegu, Korea, Republic of
Daejeon, Korea, Republic of
Incheon, Korea, Republic of
Seoul, Korea, Republic of Collapse << |
| NCT02235519 | Hypertension
... more >>Obesity
Type 2 Diabetes Mellitus Collapse << | Phase 2
Phase 3 | Unknown | June 2015 | Mexico
... more >> Hospital General de Mexico "Dr. Eduardo Liceaga"
Recruiting
Mexico, Mexico, D.F., Mexico, 06720
Contact: Juan A Peralta, bachelor Collapse << |
| NCT02756819 | - | - | Completed | - | - |
| NCT02451150 | Pediatric Hypertension | Phase 3 | Completed | - | Japan
... more >>
Fuchu, Japan
Oofu, Japan
Setagaya-ku, Japan Collapse << |
| NCT03652792 | Bioequivalence of Two Azilsart... more >>an Formulations Collapse << | Phase 1 | Completed | - | Hong Kong
... more >> Phase I Clinical Trial Centre, Chinese University of Hong Kong
Hong Kong, Sha Tin New Territories, Hong Kong, 000000 Collapse << |
| NCT02451150 | - | - | Completed | - | - |
| NCT01774591 | - | - | Completed | - | - |
| NCT02517866 | Essential Hypertension
... more >> Type 2 Diabetes Mellitus Collapse << | Phase 4 | Completed | - | China, Hong Kong
... more >>
Hong Kong, Hong Kong, China
Taiwan
Changhua county, Taiwan
Kaohsiung City, Taiwan
Kaohsiung, Taiwan
Taichung, Taiwan
Tainan City, Taiwan
Taipei, Taiwan
Taoyuan County, Taiwan
Thailand
Bangkok, Thailand
Chiang Mai, Thailand
Khon Kaen, Thailand
Pathumtani, Thailand Collapse << |
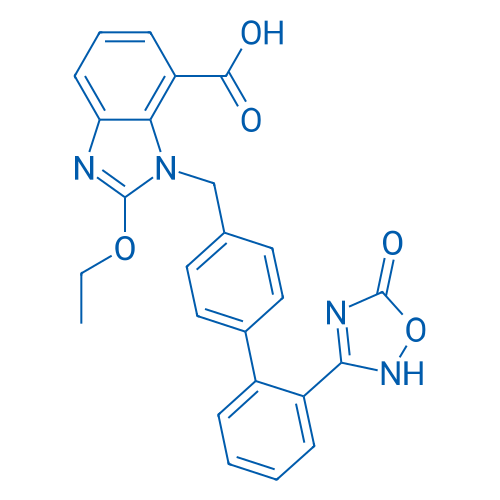

 400-920-2911
400-920-2911 sales@csnpharm.cn
sales@csnpharm.cn tech@csnpharm.cn
tech@csnpharm.cn
